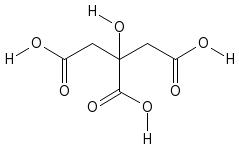
Citric Acid

Compound Name: Citric acid
Chemical Formula: C6H8O7
Molar Mass: 192.027 g/mol
Density: 1.665 ×103 kg/m3
Percent composition by mass: Hydrogen 4%, Oxygen 58%, Carbon 38%
Percent composition by number: Carbon 29%, Hydrogen 38%, Oxygen 33%
Covalent bonding
History: Citric acid was discovered in the 8th century by a Persian alchemist named Jamir ibn Hayyan. Medieval European scholars were aware of the acidity of citrus fruits. It was first isolated in 1784 by a Swedish chemist named Carl Wilhelm Scheele.
Melting Point: 153 degrees celcius
Boiling point: 157 degrees celcius
Citric acid is found in vegetables and fruits, especially citric fruits like oranges, lemons, limes, and grapefruits. Up to 8% of the dry weight of lemons and limes can be citric acid. Citric acid was first obtained by Scheele when he crystallized it from lemon juice. In 1893, C. Wehmer found that penicillium mold could produce the acid from sugar and then in 1917 an American chemist named James Currie found that some strains of aspergillus niger worked better to produce citric acid. This method is still widely used. In it, "cultures of A. niger are fed on a sucrose or glucose-containing medium to produce citric acid...After the mold is filtered out of the resulting solution, citric acid is isolated by precipitating it with lime (calcium hydroxide) to yield calcium citrate salt, from which citric acid is regenerated by treatment with sulfuric acid." (Wikipedia).
Citric acid exists as a white crystalline powder at room temperature. It exists in an anhydrous form or it can be a monohydrate. In its anhydrous form, the acid crystallizes from hot water, and as a monohydrate it crystallizes from cold water. It shares to properties of other carboxylic acids. When it is heated above 175 degrees celicius, it loses carbon dioxide and water and decomposes.
It dissolves rust off of metal objects such as copper and brass and can be used as a bleach
The chemical structure is shown below:
Chemical Formula: C6H8O7
Molar Mass: 192.027 g/mol
Density: 1.665 ×103 kg/m3
Percent composition by mass: Hydrogen 4%, Oxygen 58%, Carbon 38%
Percent composition by number: Carbon 29%, Hydrogen 38%, Oxygen 33%
Covalent bonding
History: Citric acid was discovered in the 8th century by a Persian alchemist named Jamir ibn Hayyan. Medieval European scholars were aware of the acidity of citrus fruits. It was first isolated in 1784 by a Swedish chemist named Carl Wilhelm Scheele.
Melting Point: 153 degrees celcius
Boiling point: 157 degrees celcius
Citric acid is found in vegetables and fruits, especially citric fruits like oranges, lemons, limes, and grapefruits. Up to 8% of the dry weight of lemons and limes can be citric acid. Citric acid was first obtained by Scheele when he crystallized it from lemon juice. In 1893, C. Wehmer found that penicillium mold could produce the acid from sugar and then in 1917 an American chemist named James Currie found that some strains of aspergillus niger worked better to produce citric acid. This method is still widely used. In it, "cultures of A. niger are fed on a sucrose or glucose-containing medium to produce citric acid...After the mold is filtered out of the resulting solution, citric acid is isolated by precipitating it with lime (calcium hydroxide) to yield calcium citrate salt, from which citric acid is regenerated by treatment with sulfuric acid." (Wikipedia).
Citric acid exists as a white crystalline powder at room temperature. It exists in an anhydrous form or it can be a monohydrate. In its anhydrous form, the acid crystallizes from hot water, and as a monohydrate it crystallizes from cold water. It shares to properties of other carboxylic acids. When it is heated above 175 degrees celicius, it loses carbon dioxide and water and decomposes.
It dissolves rust off of metal objects such as copper and brass and can be used as a bleach
The chemical structure is shown below: